Field Observation Report : May 11,2013
Location : HL,MO
Winter wheat : Powdery Mildew
Septoria leaf blotch
Wheat Yellow mosaic and Wheat soil-borne mosaic
Fungicide : Quilt Xcel 2.2 SE which has
propicoazole 11.7% systemmic, xylem mobile upward translocation.
mode of action: inhibit cell wall formation.
control : Eryiphe graminis
Leptoshaeria nodorum
Pseudocerosporella herpotrichoides :Eyespot = foot rot, strawbreaker
Puccinia spp.:Leaf rust = brown rust,Stripe rust
Pyrenophora teres
Rhynchosporium secalis
Septoria spp.: Septoria blotch, Stagonospora blotch
longevity: aerobic soil 30-112 days; half-life 95 days.
soil depth : residue in an acid soil deep 2-3 inches, alkali soil 8-10 inches.
azoxystrobin 13.5 % translaminar; take a few days to cover both sides.
contact fungicide; does not leach , degrade by light and soil microbes.
half-life 11 - 17 days
excellent as preventive fungicide. They effectively kill germinating spore not mycelium.
mode of action : interfere electron transportation in quinol oxidation which is " site-specific fungicide". Easy to cause mutant and lead to build up a fungicide- resistant pathogen sub population.
Suggestion:
Some diseases can cause damages beyond an economic yield loss threshold.
With this fungicide, soil-borne diseases can not be controlled and it will not decrease the source of the pathogens. The accumulation of the diseases is still in the soil, waiting for the next season to come. Need to apply the fungicide that can kill soil-borne diseases which can move downward.(pholem mobile)
Location : HL,MO
Winter wheat : Powdery Mildew
Septoria leaf blotch
Wheat Yellow mosaic and Wheat soil-borne mosaic
Fungicide : Quilt Xcel 2.2 SE which has
propicoazole 11.7% systemmic, xylem mobile upward translocation.
mode of action: inhibit cell wall formation.
control : Eryiphe graminis
Leptoshaeria nodorum
Pseudocerosporella herpotrichoides :Eyespot = foot rot, strawbreaker
Puccinia spp.:Leaf rust = brown rust,Stripe rust
Pyrenophora teres
Rhynchosporium secalis
Septoria spp.: Septoria blotch, Stagonospora blotch
longevity: aerobic soil 30-112 days; half-life 95 days.
soil depth : residue in an acid soil deep 2-3 inches, alkali soil 8-10 inches.
azoxystrobin 13.5 % translaminar; take a few days to cover both sides.
contact fungicide; does not leach , degrade by light and soil microbes.
half-life 11 - 17 days
excellent as preventive fungicide. They effectively kill germinating spore not mycelium.
mode of action : interfere electron transportation in quinol oxidation which is " site-specific fungicide". Easy to cause mutant and lead to build up a fungicide- resistant pathogen sub population.
Suggestion:
Some diseases can cause damages beyond an economic yield loss threshold.
With this fungicide, soil-borne diseases can not be controlled and it will not decrease the source of the pathogens. The accumulation of the diseases is still in the soil, waiting for the next season to come. Need to apply the fungicide that can kill soil-borne diseases which can move downward.(pholem mobile)

Septoria leaf blotch caused by fungi (.Mycosphaerella graminicola (Septoria tritici)
Hosts: Mainly wheat, but also occasionally on rye, triticale and some grass species.
Symptoms: Symptoms of Septoria can be seen very early in the growing season in most years. On young autumn-sown wheat, water-soaked patches which quickly turn brown and necrotic may be evident by early December and throughout the winter on the lowest leaves. These contain the visible black pycnidia which are the most characteristic feature of M. graminicola. Pycnidia are particularly common on dead overwintering leaves of winter wheat. Lesions on the mature plant are brown and are sometimes restricted by veins giving rectangular appearance. The black pycnidia become visible in the lesions as the symptoms develop. Lesions may coalesce leading to large areas of necrotic brown tissue.
Hosts: Mainly wheat, but also occasionally on rye, triticale and some grass species.
Symptoms: Symptoms of Septoria can be seen very early in the growing season in most years. On young autumn-sown wheat, water-soaked patches which quickly turn brown and necrotic may be evident by early December and throughout the winter on the lowest leaves. These contain the visible black pycnidia which are the most characteristic feature of M. graminicola. Pycnidia are particularly common on dead overwintering leaves of winter wheat. Lesions on the mature plant are brown and are sometimes restricted by veins giving rectangular appearance. The black pycnidia become visible in the lesions as the symptoms develop. Lesions may coalesce leading to large areas of necrotic brown tissue.
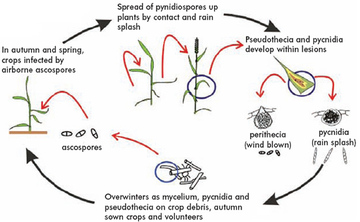
The disease cycle of M. graminicola is similar to that of S.nodorum, although M. graminicola can go through its life cycle at slightly lower temperatures (15-20°C optimum), and requires longer periods of high humidity to initiate infection. The lower leaves of winter sown crops are normally infected by long distance spread of air-borne ascospores throughout the winter and early spring. In the spring the lower leaves of the most susceptible varieties are infected and have actively sporulating lesions. Most disease spread to upper leaves occurs by rain-splash from the lower leaves which occurs during heavy rainfall. Physical spread can occur without heavy rainfall, particularly when leaves 3 and 4 overlap the upper leaves as they emerge.
Importance
The disease is the most important foliar disease on winter wheat in the UK. Losses of 50% have been reported in severely affected crops. This is largely because of the predominance of varieties which are susceptible to the disease.
Cultural control
There are no practical cultural control measures for S.tritici although date of drilling has a marked effect on disease establishment. Early drilled crops are exposed to incoming ascospores for longer periods and hence tend to have higher levels disease through the winter and early spring period. Later drilled crops may carry lower levels of disease through the winter period but this has little effect on the final level of disease in the crop as inoculum is rarely limiting, the final level of disease being determined largely by weather conditions during stem extension.
Importance
The disease is the most important foliar disease on winter wheat in the UK. Losses of 50% have been reported in severely affected crops. This is largely because of the predominance of varieties which are susceptible to the disease.
Cultural control
There are no practical cultural control measures for S.tritici although date of drilling has a marked effect on disease establishment. Early drilled crops are exposed to incoming ascospores for longer periods and hence tend to have higher levels disease through the winter and early spring period. Later drilled crops may carry lower levels of disease through the winter period but this has little effect on the final level of disease in the crop as inoculum is rarely limiting, the final level of disease being determined largely by weather conditions during stem extension.

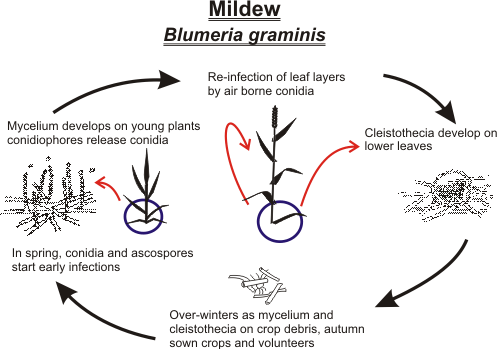
Powdery Mildew : Blumeria. graminis f. sp tritici affects wheat and triticale
Mildew overwinters primarily as mycelium on volunteers and autumn-sown crops. The cleistothecia produced during late summer are resistant to low temperatures and drying out, and they allow the fungus to survive for a time in the absence of a host. In humid weather, cleistothecia release the sexually produced ascospores which can initiate autumn infections. As temperatures rise in the spring, dormant mycelium starts to grow and spores are quickly produced. These germinate over a wide range of temperatures, from 5OC to 30OC, although 15OC is optimal with relative humidity above 95%. Free water inhibits spore germination. Under dry conditions, fresh spores can be formed in about 7 days. At the end of the season, volunteers and early autumn-sown crops may become infected, providing inoculum for the following crop.
Importance
Late-sown winter wheat crops are often particularly prone to attack, especially when growing rapidly in the spring. Excessive nitrogen fertiliser also encourages the disease and mildew can be particularly severe in dense crops. The visual appearance of the disease usually outweighs its damage potential especially during the autumn and winter. In susceptible varieties yield losses can be high (up to 20%) and early control can be very important. However, the disease generally causes much smaller yield losses and late attacks (after flowering) on the flag-leaf and ear rarely cause significant losses.
Cultural control
The eradication of volunteer cereals which act as overwintering sources of inoculum and disposal of stubble and debris which may be infested with cleistothecia can be important in mildew control. High levels of nitrogen fertiliser should be avoided. Later drillings of wheat tend to suffer more from mildew attack.
Wheat varieties exhibit a wide range of resistance to the disease and many current varieties have good disease resistance. However the range of sources of resistance used in most current wheat varieties is relatively narrow. Some varieties from other European countries can be very prone to mildew attack.
Life cycle
Mildew overwinters primarily as mycelium on volunteers and autumn-sown crops. The cleistothecia produced during late summer are resistant to low temperatures and drying out, and they allow the fungus to survive for a time in the absence of a host. In humid weather, cleistothecia release the sexually produced ascospores which can initiate autumn infections. As temperatures rise in the spring, dormant mycelium starts to grow and spores are quickly produced. These germinate over a wide range of temperatures, from 5OC to 30OC, although 15OC is optimal with relative humidity above 95%. Free water inhibits spore germination. Under dry conditions, fresh spores can be formed in about 7 days. At the end of the season, volunteers and early autumn-sown crops may become infected, providing inoculum for the following crop.
Importance
Late-sown winter wheat crops are often particularly prone to attack, especially when growing rapidly in the spring. Excessive nitrogen fertiliser also encourages the disease and mildew can be particularly severe in dense crops. The visual appearance of the disease usually outweighs its damage potential especially during the autumn and winter. In susceptible varieties yield losses can be high (up to 20%) and early control can be very important. However, the disease generally causes much smaller yield losses and late attacks (after flowering) on the flag-leaf and ear rarely cause significant losses.
Cultural control
The eradication of volunteer cereals which act as overwintering sources of inoculum and disposal of stubble and debris which may be infested with cleistothecia can be important in mildew control. High levels of nitrogen fertiliser should be avoided. Later drillings of wheat tend to suffer more from mildew attack.
Wheat varieties exhibit a wide range of resistance to the disease and many current varieties have good disease resistance. However the range of sources of resistance used in most current wheat varieties is relatively narrow. Some varieties from other European countries can be very prone to mildew attack.
Life cycle

WHEAT YELLOW MOSAIC (WHEAT SPINDLE STREAK MOSAIC)
Wheat yellow mosaic (usually called wheat spindle streak mosaic) is caused by a soilborne virus which also is transmitted by the soilborne fungus, Polymyxa graminis. The virus can survive for 10 years or more in soil in close association with the fungus. Wheat yellow mosaic apparently makes plants resistant to soilborne mosaic virus. The ratio of soilborne wheat mosaic virus to wheat yellow mosaic virus in plants infected with both viruses is about 20:1.
The disease is found in Illinois in the general area where soilborne wheat mosaic is prevalent. In southwestern Ontario, yield losses occur each year and may reach 40 percent in some fields where very susceptible cultivars are grown. The only host for wheat yellow mosaic is wheat, except in Germany, where it has been reported on barley and rye. Like soilborne wheat mosaic, both winter and spring wheats are susceptible to the virus but spring wheats rarely develop symptoms.
Symptoms:
The first leaves produced in early spring develop yellow-green mottling, dashes, and streaks. The discontinuous streaks are oriented parallel with the leaf veins and taper at each end to form yellowish"spindles" . Symptoms are most prominent on the lower leaves because warmer spring temperatures present their development on younger leaves. As the leaves mature and when temperatures remain cool, the center of the spindle may turn brown, streaking may progress to the flag leaf, and the yellow-green areas tend to merge. Reddish streaking and dieback of leaf tips or entire leaves sometimes .
occurs. Infected wheat plants remain slightly stunted and produce fewer tillers than healthy plants. When warm weather arrives, new symptomless leaves hide the lower leaves showing symptoms. Fewer head sand kernels are produced on infected plants, but kernel weight is not appreciably affected. Cold hardiness is reduced by infection with the virus. The disease tends to be more uniformly distributed through out fields than soilborne wheat mosaic
Wheat yellow mosaic (usually called wheat spindle streak mosaic) is caused by a soilborne virus which also is transmitted by the soilborne fungus, Polymyxa graminis. The virus can survive for 10 years or more in soil in close association with the fungus. Wheat yellow mosaic apparently makes plants resistant to soilborne mosaic virus. The ratio of soilborne wheat mosaic virus to wheat yellow mosaic virus in plants infected with both viruses is about 20:1.
The disease is found in Illinois in the general area where soilborne wheat mosaic is prevalent. In southwestern Ontario, yield losses occur each year and may reach 40 percent in some fields where very susceptible cultivars are grown. The only host for wheat yellow mosaic is wheat, except in Germany, where it has been reported on barley and rye. Like soilborne wheat mosaic, both winter and spring wheats are susceptible to the virus but spring wheats rarely develop symptoms.
Symptoms:
The first leaves produced in early spring develop yellow-green mottling, dashes, and streaks. The discontinuous streaks are oriented parallel with the leaf veins and taper at each end to form yellowish"spindles" . Symptoms are most prominent on the lower leaves because warmer spring temperatures present their development on younger leaves. As the leaves mature and when temperatures remain cool, the center of the spindle may turn brown, streaking may progress to the flag leaf, and the yellow-green areas tend to merge. Reddish streaking and dieback of leaf tips or entire leaves sometimes .
occurs. Infected wheat plants remain slightly stunted and produce fewer tillers than healthy plants. When warm weather arrives, new symptomless leaves hide the lower leaves showing symptoms. Fewer head sand kernels are produced on infected plants, but kernel weight is not appreciably affected. Cold hardiness is reduced by infection with the virus. The disease tends to be more uniformly distributed through out fields than soilborne wheat mosaic
Wheat soil-borne mosaic vector by Polymyxa graminis . This disease caused by protozoan's zoospore.(Fungus-like)
Alternative Host : fall-sown rye, barley, emmer, and spelt,wild annual bromegrass (Bromus commutatus), sorghum, and some species of Chenopodium .
Transmission : infectious in dried leaves for several years or more. It is not transmitted through the seed or by insects, but it is transmitted by a soil-inhabiting fungus or mechanically at low rates.
Symptoms: chlorotic leaf mottling or mosaic and rosetting or stunting.
Plants infected with the soil borne wheat mosaic virus usually appear in early spring as irregular patches of light green to bronze-yellow or light purple wheat within a field – depending on the cultivar, strain of the virus, and seasonal growing conditions.
The disease often occurs in poorly drained low areas and waterways in fields. A mosaic-infected area does not increase in size during the growing season. The infected area may increase over time due to tillage,land leveling, or flooding of fields.
Under conditions unfavorable for growth, however,infected plants remain dwarfed to maturity. Roots may be more severely stunted than shoots in some cultivars. Some plants may die, while others will produce fewer stems (culms) and heads. Maturity is often delayed. The heads on diseased plants may be shorter than normal heads and have shriveled, lightweight kernels.
The severity of this disease varies greatly, depending on the resistance of the wheat cultivar, concentration and virulence of the virus strain(s) in the soil, weather conditions, and planting date. A prolonged, cool growing period with a mean temperature below 60°F (16°C) appears necessary before susceptible wheat cultivars are appreciably damaged by this disease.
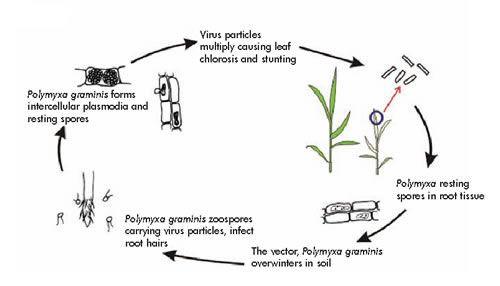
Disease Cycle: The virus causing soilborne wheat mosaic survives in the soil and crop residues from season to season protected by its fungal vector or agent, Polymyxa graminis, an obligate parasite in the roots of many grasses and a few higher plants. During cool wet periods motile spores (zoospores) are released by the fungus and infect roots of wheat plants. The virus particles are either inside the spores of the fungus (both zoospores and thick-walled resting spores) or tightly bound to the surface of these spores. The fungus normally infects wheat roots shortly after planting in the fall. The symptoms, however, do not normally appear until early spring.
The virus is transmitted from plant to plant by its fungal vector. The zoospores are produced in virus-infected roots or debris and carry the virus. The zoospores swim through the soil solution to healthy root hairs and epidermal cells, penetrate them, and thus inoculate the plant. Once inside the plant, P. graminis replaces plant cell contents with plasmodial bodies that either segment into additional zoospores or develop into resting spores two to four weeks after infection.The Polymyxa fungus survives unfavorable periods in the form of resting spores clustered in the cortical and epidermal cells within plant debris . These spores can survive in soil for 10 years or more in the absence of wheat or other host plants. The virus and its transmitting fungus are capable of spreading with any movement of infested soil, even wind-blown dust. Because the swimming zoospores of the fungus transmit the virus, soilborne wheat mosaic is most common and severe in low, wet areas of fields in years when fall rainfall is ample.
References: www.hgca.com
www.ncbi.nlm.nih.gov/pubmed/15242168
www.ars.usda.gov/SP2UserFiles/ad_hoc/.../CowgerWeisz05.pdf
wheat.pw.usda.gov/ggpages/wheatpests.html
Pictures: CE7Plus
Alternative Host : fall-sown rye, barley, emmer, and spelt,wild annual bromegrass (Bromus commutatus), sorghum, and some species of Chenopodium .
Transmission : infectious in dried leaves for several years or more. It is not transmitted through the seed or by insects, but it is transmitted by a soil-inhabiting fungus or mechanically at low rates.
Symptoms: chlorotic leaf mottling or mosaic and rosetting or stunting.
Plants infected with the soil borne wheat mosaic virus usually appear in early spring as irregular patches of light green to bronze-yellow or light purple wheat within a field – depending on the cultivar, strain of the virus, and seasonal growing conditions.
The disease often occurs in poorly drained low areas and waterways in fields. A mosaic-infected area does not increase in size during the growing season. The infected area may increase over time due to tillage,land leveling, or flooding of fields.
Under conditions unfavorable for growth, however,infected plants remain dwarfed to maturity. Roots may be more severely stunted than shoots in some cultivars. Some plants may die, while others will produce fewer stems (culms) and heads. Maturity is often delayed. The heads on diseased plants may be shorter than normal heads and have shriveled, lightweight kernels.
The severity of this disease varies greatly, depending on the resistance of the wheat cultivar, concentration and virulence of the virus strain(s) in the soil, weather conditions, and planting date. A prolonged, cool growing period with a mean temperature below 60°F (16°C) appears necessary before susceptible wheat cultivars are appreciably damaged by this disease.
Disease Cycle: The virus causing soilborne wheat mosaic survives in the soil and crop residues from season to season protected by its fungal vector or agent, Polymyxa graminis, an obligate parasite in the roots of many grasses and a few higher plants. During cool wet periods motile spores (zoospores) are released by the fungus and infect roots of wheat plants. The virus particles are either inside the spores of the fungus (both zoospores and thick-walled resting spores) or tightly bound to the surface of these spores. The fungus normally infects wheat roots shortly after planting in the fall. The symptoms, however, do not normally appear until early spring.
The virus is transmitted from plant to plant by its fungal vector. The zoospores are produced in virus-infected roots or debris and carry the virus. The zoospores swim through the soil solution to healthy root hairs and epidermal cells, penetrate them, and thus inoculate the plant. Once inside the plant, P. graminis replaces plant cell contents with plasmodial bodies that either segment into additional zoospores or develop into resting spores two to four weeks after infection.The Polymyxa fungus survives unfavorable periods in the form of resting spores clustered in the cortical and epidermal cells within plant debris . These spores can survive in soil for 10 years or more in the absence of wheat or other host plants. The virus and its transmitting fungus are capable of spreading with any movement of infested soil, even wind-blown dust. Because the swimming zoospores of the fungus transmit the virus, soilborne wheat mosaic is most common and severe in low, wet areas of fields in years when fall rainfall is ample.
References: www.hgca.com
www.ncbi.nlm.nih.gov/pubmed/15242168
www.ars.usda.gov/SP2UserFiles/ad_hoc/.../CowgerWeisz05.pdf
wheat.pw.usda.gov/ggpages/wheatpests.html
Pictures: CE7Plus

 RSS Feed
RSS Feed
